For decades, leukemia and lymphoma were treated with one approach: chemotherapy. It was brutal, unpredictable, and often left patients weaker than before. But today, a quiet revolution is underway. Targeted and cellular therapies are changing the game-offering more precision, fewer side effects, and in some cases, real cures where none existed before.
What Exactly Are Targeted Therapies?
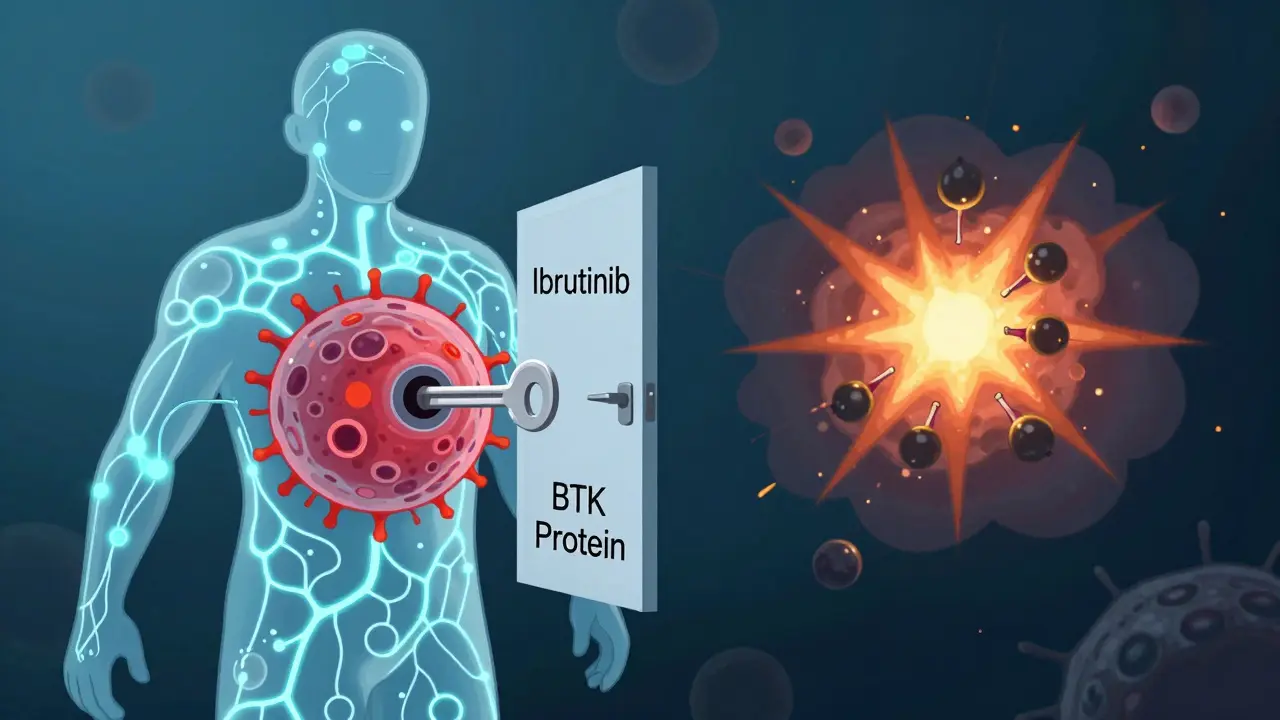
Targeted therapies don’t attack all fast-growing cells like chemo does. They go after specific molecular weak spots in cancer cells. Think of it like using a key to unlock a single door instead of blowing up the whole building. For chronic lymphocytic leukemia (CLL) and small lymphocytic lymphoma (SLL), two drugs have become standard: ibrutinib (Imbruvica) and venetoclax (Venclexta). Ibrutinib blocks a protein called Bruton’s tyrosine kinase (BTK), which leukemia cells need to survive. It’s taken as a pill, once a day. Venetoclax goes after BCL-2, a protein that keeps cancer cells from dying. Together, they can push the disease into deep remission. The big win? These drugs are oral, meaning patients don’t need hospital stays. A 2025 study from the CLL Society found that patients on targeted therapies took nearly twice as long to develop Richter transformation-a dangerous leap into aggressive lymphoma-compared to those on old-school chemo. The median time jumped from 2.2 years to 4.9 years. That’s not just a statistic. That’s extra years of working, traveling, watching your kids grow up.How CAR T-Cell Therapy Works
If targeted therapies are like precision missiles, CAR T-cell therapy is like training your own immune system to become a living weapon. Here’s how it works: First, doctors pull out your T cells-your body’s natural assassins-through a process called leukapheresis. Then, in a lab, those cells are genetically rewired to carry a special receptor called a chimeric antigen receptor (CAR). This CAR is designed to latch onto CD19, a protein found on most B-cell lymphomas and leukemias. The modified cells are multiplied, then infused back into you. The result? A living army of T cells that hunt down cancer like a GPS-guided missile. The first approved CAR T, tisagenlecleucel (Kymriah), was approved in 2017 for kids with relapsed acute lymphoblastic leukemia. Today, we have newer versions like axicabtagene ciloleucel (Yescarta) and lisocabtagene maraleucel (liso-cel). The numbers are staggering. In a 2025 trial for relapsed mantle cell lymphoma, a new CAR T called LV20.19 achieved a 100% response rate-with 88% of patients achieving complete remission. In another study, a dual-target CAR T that hits both CD19 and CD20 saw a 63.6% complete remission rate in patients who had failed every other treatment. For some, this is the first time they’ve been cancer-free in years.Targeted vs. Cellular: What’s the Difference?
It’s not a competition. They’re different tools for different jobs.| Feature | Targeted Therapies | Cellular Therapies (CAR T) |
|---|---|---|
| Administration | Oral pills, daily | Single IV infusion |
| Duration | Lifelong (unless stopped for resistance) | One-time treatment |
| Response Time | Weeks to months | Days to weeks |
| Major Toxicity | Bleeding, diarrhea, fatigue | Cytokine release syndrome, neurotoxicity |
| Cost (2025 avg) | $15,000-$25,000/month | $373,000-$475,000 total |
| Best For | Chronic, stable disease | Relapsed/refractory disease |
Targeted therapies are ideal for long-term control. Many patients stay on them for years. But resistance eventually builds. A 2025 review in the Journal of Clinical Oncology showed that BTK inhibitors typically work for 3-5 years before the cancer finds a way around them.
CAR T-cell therapy, on the other hand, is a one-shot deal. If it works, the immune cells stick around-sometimes for life. That’s why it’s reserved for patients who’ve tried everything else. But it’s also why it’s becoming a first-line option for high-risk cases. By 2030, 68% of hematologists predict CAR T will be used upfront for aggressive lymphomas.
Why This Isn’t Perfect Yet
These therapies are miracles-but they’re not magic. CAR T-cell therapy can trigger cytokine release syndrome (CRS), where the immune system goes haywire. Symptoms include high fever, low blood pressure, and trouble breathing. Neurotoxicity can follow, causing confusion, seizures, or even coma. About 20-40% of patients experience these side effects. That’s why it’s only given in certified centers with ICU access. Setting up a CAR T program takes 6-12 months and costs millions. Targeted drugs aren’t risk-free either. Venetoclax can cause tumor lysis syndrome-when dying cancer cells flood the bloodstream with toxins. That’s why patients start on low doses and are monitored in the hospital during the ramp-up. And then there’s cost. A single CAR T treatment can hit $400,000. Even with insurance, out-of-pocket costs can hit $15,000-$25,000 a month for targeted therapies. That’s why some doctors say we’re facing an ethical crisis: Who gets access when the price tag is so high?What’s Next? The Future Is Here
The next wave of therapies is already in the lab. Gilead’s Kite division is testing dual-target CAR T-cells like KITE-363 and KITE-753. These don’t just go after CD19-they also hit CD20. Why? Because cancer cells sometimes drop CD19 to escape treatment. With two targets, they can’t hide. Early data shows fewer relapses and less severe side effects. Some experts think this could make CAR T safe enough to give outside a hospital one day. New BTK inhibitors are also coming. Acalabrutinib (Calquence) is already in use, but next-gen versions aim to be more selective, cutting down on side effects like bleeding and atrial fibrillation. And then there’s the big question: Can we move CAR T earlier in treatment? Right now, it’s a last resort. But in 2025, the FDA gave priority review to liso-cel for marginal zone lymphoma-a sign that these therapies are expanding beyond the most desperate cases.
What Patients Need to Know
If you or someone you love has leukemia or lymphoma, here’s what matters:- Targeted therapies are great for long-term control, especially if you’re not a candidate for intensive treatment.
- CAR T-cell therapy is not a last-ditch effort anymore-it’s a real chance at cure, even after multiple relapses.
- Not every center can give CAR T. Ask if your hospital is certified by the FDA or NCI.
- Resistance to BTK inhibitors is common. If your cancer returns, don’t assume there’s nothing left. Clinical trials are expanding fast.
- Cost is a real barrier. Talk to your care team about financial aid programs. Many manufacturers offer patient support services.
There’s no one-size-fits-all anymore. A 72-year-old with CLL might do better on venetoclax and ibrutinib. A 35-year-old with relapsed lymphoma might have a shot at life-long remission with CAR T. The key is knowing your options-and having a team that knows how to use them.
Are targeted therapies better than chemotherapy for leukemia and lymphoma?
Yes, for most patients. Targeted therapies like ibrutinib and venetoclax are more effective, cause fewer side effects, and don’t require hospitalization. Studies show they extend progression-free survival and reduce complications like infections and organ damage that often follow chemo. They’re now the standard for CLL and many lymphomas, replacing older chemoimmunotherapy regimens.
Can CAR T-cell therapy cure lymphoma?
For some patients, yes. In relapsed or refractory large B-cell lymphoma, CAR T-cell therapy like Yescarta has shown 4-year survival rates of over 40%-a number unheard of just a decade ago. In mantle cell lymphoma, some patients achieve complete remission that lasts for years. While not every patient is cured, a significant portion live cancer-free without needing further treatment.
Why is CAR T-cell therapy so expensive?
It’s a personalized, one-time treatment made in a lab using your own cells. Each batch is custom-built, requiring specialized facilities, trained staff, and weeks of manufacturing. The process includes leukapheresis, genetic engineering, cell expansion, quality testing, and infusion-all under strict regulations. Plus, companies recover R&D costs from a small patient pool. The average cost is $373,000-$475,000, but financial assistance programs are widely available.
Do these therapies work for all types of leukemia and lymphoma?
No. Targeted therapies are most effective in B-cell cancers like CLL, SLL, follicular lymphoma, and mantle cell lymphoma. CAR T-cell therapy is approved for certain B-cell acute lymphoblastic leukemias and aggressive B-cell lymphomas. They don’t work for T-cell lymphomas or most AML cases-though trials are underway. Testing for specific markers like CD19, CD20, or BTK mutations is required before treatment.
What happens if targeted therapy stops working?
It’s common. Resistance often develops after 3-5 years. But that doesn’t mean there’s nothing left. Doctors can switch to another targeted drug, combine therapies, or move to CAR T-cell therapy. Trials are testing next-gen BTK inhibitors and drugs that target alternative pathways like PI3K or BCL-2. For patients with TP53 mutations, CAR T is often the best next step.
Is CAR T-cell therapy available everywhere?
No. Only about 89% of NCI-designated cancer centers offer it, and just 32% of community hospitals do. The reason? It requires specialized labs, ICU support, and staff trained in managing severe side effects like cytokine release syndrome. Patients often need to travel to a major center. Insurance usually covers it if you meet clinical criteria, but logistics can take weeks to arrange.
Final Thoughts
We’re no longer just trying to kill cancer. We’re learning to outsmart it. Targeted therapies give patients control. CAR T-cell therapy gives them hope. Together, they’ve turned once-fatal diseases into manageable ones-and in some cases, curable ones. The challenge now isn’t just science. It’s access. It’s cost. It’s making sure these breakthroughs don’t just help the lucky few, but everyone who needs them.For those facing leukemia or lymphoma today, the message is clear: There are more options than ever. And the future isn’t just about surviving-it’s about living.

Comments (9)
Pankaj Gupta
March 1, 2026 AT 16:57Targeted therapies represent a paradigm shift in oncology-not merely an incremental improvement. The precision of BTK and BCL-2 inhibitors fundamentally alters the risk-benefit calculus for patients with CLL and SLL. Unlike chemotherapy, which indiscriminately assaults proliferating cells, these agents exploit molecular vulnerabilities unique to malignant B-cells. This specificity translates into tangible quality-of-life gains: fewer hospitalizations, reduced neutropenia, and preserved organ function. The data from the CLL Society is compelling, but we must also acknowledge the psychological impact of oral therapy-patients retain agency, autonomy, and routine. These are not just clinical endpoints; they are human ones.
Moreover, the emergence of resistance mechanisms underscores the necessity of longitudinal monitoring and adaptive treatment strategies. The 3-5 year window for BTK inhibitors is not a failure-it is a phase of disease evolution that demands smarter sequencing, not abandonment.
Future research must prioritize biomarkers of resistance beyond BTK mutations: mutations in PLCG2, CARD11, and TNFRSF14 are increasingly relevant. We are moving from empiric treatment to predictive oncology.
Alex Brad
March 2, 2026 AT 19:38CAR T is revolutionary, but it’s not a cure-all. The toxicity profile still scares off many patients and providers. We need better management protocols and earlier access-not just for the desperate, but for those who could benefit before they’re too far gone.
Renee Jackson
March 3, 2026 AT 02:04As a clinical nurse specialist working in hematology, I can attest to the transformative impact of these therapies on patient outcomes. The shift from chemotherapy to targeted agents has allowed individuals to maintain employment, attend their children’s graduations, and travel to see loved ones-things that were once unthinkable after a diagnosis of advanced lymphoma.
CAR T-cell therapy, while complex and resource-intensive, offers a rare opportunity for durable remission in patients who had exhausted all other options. The fact that some achieve complete remission lasting over five years is nothing short of miraculous. However, we must ensure that access is equitable and that every patient, regardless of zip code or socioeconomic status, has the opportunity to benefit from these advances. Support systems, financial counseling, and transportation assistance are not luxuries-they are essential components of care.
Levi Viloria
March 3, 2026 AT 12:36It’s wild how much has changed in just ten years. I remember when chemo was the only option-people would lose their hair, get sick constantly, and still not beat the cancer. Now you’ve got pills you can take at home and a one-time infusion that trains your immune system to hunt down cancer like a robot ninja. The science is insane.
But honestly? The cost still feels like a joke. $400K for one treatment? That’s a house. Or a car. Or a decade of rent. And don’t get me started on how few hospitals can even do CAR T. You’ve got people flying across the country just to get a shot at life. It’s progress, sure-but it’s also a system that’s still rigged for the wealthy.
Richard Elric5111
March 4, 2026 AT 02:51One cannot help but reflect upon the ontological implications of these therapeutic modalities. The targeted agent, in its molecular specificity, embodies a reductionist epistemology of disease: cancer as a malfunctioning circuit, amenable to correction through pharmacological intervention. Yet CAR T-cell therapy transcends this framework-it is not merely a drug, but an engineered biological entity, a living pharmacopeia derived from the patient’s own somatic cells.
Thus, we are no longer treating a disease; we are reconstituting the immune self. This blurs the boundary between therapy and identity. Is the patient who receives CAR T the same person after infusion? Or has their biological essence been irrevocably altered? These are not rhetorical questions-they are the philosophical underpinnings of modern oncology.
Dean Jones
March 5, 2026 AT 02:07Let’s cut through the corporate hype. Yes, targeted therapies and CAR T are amazing. But let’s not pretend this is a revolution that’s helping everyone. The truth? These treatments are designed for patients who are already privileged-white, insured, educated, and geographically proximate to academic medical centers. The fact that only 32% of community hospitals offer CAR T? That’s not an infrastructure gap-it’s a moral failure. The pharmaceutical industry has turned cancer into a luxury good, and the FDA has been complicit by approving drugs based on surrogate endpoints while ignoring real-world access.
And don’t get me started on the financial toxicity. A patient on ibrutinib pays $20,000 a month out of pocket? That’s not a treatment plan-it’s a debt trap. Meanwhile, the same companies that charge $475,000 for CAR T are spending millions lobbying Congress to block price controls. This isn’t science. This is capitalism with a white coat.
And yet, we celebrate these therapies like they’re miracles without asking who’s being left behind. The 72-year-old in rural Mississippi? The single mom in Detroit without transportation? The undocumented immigrant with CLL? They’re not part of the clinical trial data. They’re not in the marketing brochures. They’re invisible.
So yes, the science is brilliant. But until we fix the system that hoards access behind wealth and geography, we’re not curing cancer-we’re just making the rich live longer while everyone else dies waiting.
Betsy Silverman
March 5, 2026 AT 07:31I’ve seen firsthand how these therapies change lives-not just medically, but emotionally. A patient I worked with last year had relapsed twice after chemo. She was told her options were limited. Then she got CAR T. Six months later, she was hiking with her granddaughter. That’s the kind of moment that reminds you why we do this.
But we also need to talk about the emotional toll on caregivers. The anxiety of waiting for side effects to resolve, the stress of coordinating travel, the fear of financial ruin. We need more support systems-not just for patients, but for their families. This isn’t just about science. It’s about compassion.
Ivan Viktor
March 6, 2026 AT 13:29So we spent $400K to train your immune system to kill cancer… and now you’re still alive? Wow. Groundbreaking. Next up: turning water into wine. I’m sure the FDA will approve that too.
Zacharia Reda
March 7, 2026 AT 21:59Interesting that no one’s talking about the elephant in the room: what happens when these therapies stop working? We’re so focused on the upfront wins-remission, survival, quality of life-that we ignore the long game. Resistance is inevitable. BTK inhibitors fail. CAR T cells exhaust. Tumors evolve. And then what? We don’t have a clear roadmap for second-line options beyond ‘try another trial.’
Meanwhile, the pipeline is crowded with next-gen inhibitors and dual-target CAR Ts, but who’s funding the studies on what comes *after* those fail? We’re building a ladder to the stars, but we’re not planning for the fall.
Also, the cost argument is valid, but let’s be real: if we spent half as much on early detection and prevention as we do on these one-time miracles, we’d be saving more lives for less money. Maybe we need to stop treating cancer like a terminal emergency and start treating it like a chronic condition we can manage-before it becomes a crisis.